
Trazodone is a sedative antidepressant with a dual serotonergic mechanism of action. is authorized to major depressive episodes and for mixed states of depression-anxiety with/without secondary insomnia. Too a shown a beneficial effect in the treatment of clinical situations not included in the data sheet, such as insomnia (not associated with depression), post-traumatic stress and behavioral disorders associated with substance use, among others. It is one of the most prescribed drugs, after benzodiazepines and hypnotics.
Z (zolpidem, zopiclone and zaleplon), for the management of insomnia. As it is used outside of the technical data sheet, the
informed consent of the patient. This article collects information regarding its efficacy and safety in this
indication.
PHARMACOLOGICAL CHARACTERISTICS

Trazodone is an antidepressant belonging to the SARI group (serotonin-2 antagonist/reuptake inhibitor), it is a serotonin reuptake inhibitor and 5HT21 receptor antagonist. The activation of the latter receptor is commonly associated with insomnia, anxiety, psychomotor agitation, and changes in sexual function.
The sedative effect is believed to be due to its relatively high antagonistic affinity for the central α-1 receptor and its low affinity for H12 receptors, to which its modulating effect on cortisol suppression could also contribute through the hypothalamic-pituitary-adrenal axis.
It has a moderate antihistamine activity and low anticholinergic load3, not presenting the typical effects of tricyclic antidepressants on cardiac function and being relatively well tolerated in glaucomatous patients and with urinary retention problems. At low doses (25-100 mg) it induces and maintains sleep without causing daytime drowsiness or tolerance, thanks to its short plasma half-life (3-6 h), since at these doses it only blocks histamine 1 and α-1 receptors adrenergic. In order to achieve the antidepressant effect, the inhibition of serotonin reuptake is also required. which occurs with high doses (150-600 mg).
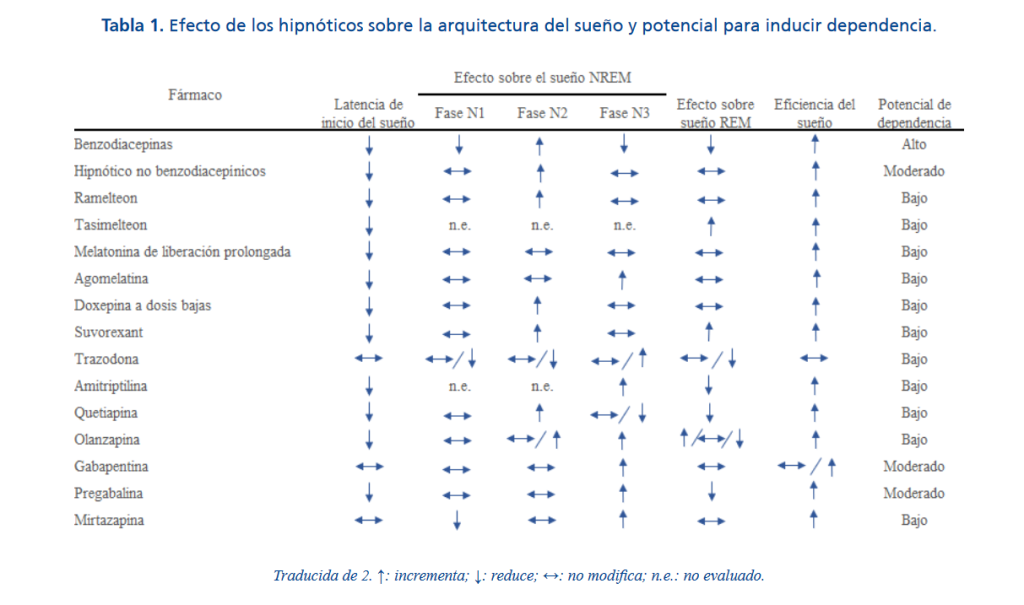
Table 1 shows the scant effect of trazodone on sleep architecture and its low capacity to produce dependence, compared to other drugs used in insomnia. Regarding the architecture of the sleep, there are two types of sleep, REM (with rapid eye movement, brain activity similar to wakefulness and completely relaxed muscles) and NREM (without rapid eye movement), which is subdivided into phase 1 (drowsiness), phase 2 (shallow sleep) and phases 3 and 4 (deep sleep). After falling asleep, the 4 o’clock NREM phases and then transition to REM. NREM and REM dreams alternate, giving rise to a sleep cycle that lasts about 100 minutes. The ideal hypnotic is one that does not affect sleep architecture.
EFFICACY IN THE TREATMENT OF INSOMNIA
The available evidence on the efficacy of trazodone in the treatment of insomnia is scant, obtained in its most small clinical trials (usually with subjects with depressive disorders) and reviews their systematics.
PRIMARY INSOMNIA
Primary insomnia is defined as sleep disturbances that last at least one month and have no specific etiology. identifiable. In this type of insomnia, the most relevant evidence comes from a study comparing trazodone 50 mg/day, zolpidem 10 mg/day, and placebo in 278 nondepressed adults suffering from sleep-conciliation primary insomnia.
At least one month in duration, characterized by a sleep latency time of 30 minutes or more, and a durationof 4-6 hours of sleep, at least three nights a week.
- After one week, trazodone, compared to placebo, reduced the onset latency by 10.2 minutes to sleep (less reduction than zolpidem) and increased sleep duration by 21.8 minutes. Both Variables were measured by the patient himself. The differences were statistically significant, butthey were not considered clinically relevant.
- At two weeks, the increase in sleep duration was maintained, although no benefit was observed in terms ofreduction of sleep onset latency time.
SECONDARY INSOMNIA
Secondary insomnia can be caused by psychiatric illnesses or certain clinical situations, environmental factors, changes in the circadian rhythm or the use of certain medications. In this type of insomnia, trazodone for a short period of time (6-12 weeks) and at low doses (50-100mg/day) decreases the sleep latency period and increases the time that the patient spends asleep, without having a significant improvement in efficacy was found, although there was a significant improvement in the patient’s perception of quality sleep.
A review published in the journal Pharmacological Reviews in 2018 includes randomized clinical trials with control group in patients with insomnia secondary to different clinical situations.
- Insomnia secondary to major depression. In a six-week clinical trial in 225 patients, in which compared trazodone (294-300 mg/day) versus venlafaxine (160 mg/day) and placebo, trazodone improved the Hamilton Depression Scale items related to insomnia, while venlafaxine improved plus those related to psychomotor inhibition. Regarding adverse effects, there was more abandonment of treatment in the trazodone group for dizziness and the venlafaxine group for nausea. In two reviews that analyzed mainly patients with major depression, it is concluded that the evidence of efficacy of trazodone is limited, despite having observed an increase in total sleep duration. This, together with the high rate ofdiscontinuation due to adverse events (sedation, dizziness, psychomotor affectation and even priapism in 1/6000 patients) makes its benefit-risk balance in this population group uncertain.
- Antidepressant-induced insomnia. Of three studies of 17, 12 and 7 patients, only two observed someefficacy relative to placebo on selected sleep parameters.
- Insomnia secondary to alcohol or opioid dependence. Its use is not recommended in these patients for not having observed efficacy, in the case of patients in treatment with methadone, and for worsening the withdrawal during alcohol detox.
Sleep disturbances are common in patients with dementia, so the use of antidepressants with effective sedative might be of interest. In a small clinical trial with 30 patients with Alzheimer’s disease, of 2 weeks of duration, an improvement in total night sleep time was observed in those treated with 50 mg/day of trazodone compared to the placebo group. Low-quality evidence is considered.
OTHER USES: COGNITIVE-BEHAVIORAL SYMPTOMS OF DEMENTIA
Finally, it should be noted that some authors consider that antidepressants could be effective for the treatment of psychological and behavioral symptoms of dementia. A systematic review collects data from three clinical trials with 28, 149 and 31 patients with dementia, disease Alzheimer’s disease and frontotemporal dementia, respectively, in which trazodone 250-300 mg/day is compared with haloperidol 5 mg/day (in two trials) and placebo (one trial). It concludes that, taking into account the risks of the antipsychotics, antidepressants could be considered an alternative in these patients. In a population-based study (BIFAP primary care database) conducted with people over 65 years of age in Spain it was observed that the consumption of trazodone had multiplied by five between 2002 and 2011, with main uses: depression (21%), dementia (20%), insomnia (16%) and anxiety (9%). In over 75 years trazodone was mainly used to treat certain symptoms associated with dementia (irritability, anxiety and verbal aggression). Despite its widespread use, they do not find sufficient evidence to support it.
SAFETY
The safety profile of trazodone is well established and documented, although it must be taken into account that most of the studies with this drug were focused on its use for the treatment of disorders depressive. In general, it is a safe drug, well tolerated and with a safety profile that differs from that of other hypnotic. The most common adverse reactions are mild: dizziness, drowsiness, fatigue, headache, dry mouth,nervousness, diarrhea and syncope, among others. Driving should be avoided during treatment.
Although for insomnia the dose used is lower than that used to treat depressive syndromes, there are Special care must be taken with the elderly population, which is more prone to suffering orthostatic hypotension due to dysfunction autonomic function due to age (greater risk of falls, especially at night)1. In this population it has been described also changes in memory, balance and muscle strength upon awakening. Regarding interactions with other drugs, it can intensify the sedative effect of antipsychotics, hypnotics, sedatives, anxiolytics and antihistamines, so in case of joint use it is recommended to use lower doses and avoid alcohol consumption during treatment. In addition, cases of serotonin syndrome have been described(confusion/agitation, diarrhoea, fever, sweating, motor incoordination) due to administration with other substances serotonergic agents, such as tricyclic antidepressants, SSRIs, SNRIs, or MAOIs), triptans, and neuroleptics. They have evendescribed neuroleptic malignant syndromes resulting in death due to administration with neuroleptics. In these cases, trazodone should be discontinued immediately and symptom support treatment initiated.
Trazodone has a not very potent anticholinergic effect and is considered one of the antidepressants with the best profile. cardiotoxic, although it has been associated with an increased risk of prolongation of the QT interval, which can cause severe arrhythmias such as torsade de pointes14,15. This risk increases with age and with the presence of comorbidities, especially cardiovascular. This association has been demonstrated especially in cases of trazodone overdose, since at therapeutic doses the evidence related to this risk is not entirely consistent. On the one hand, a study carried out with 36 healthy volunteers showed an association between the consumption of trazodone and elongation of the QT interval, without observing significant differences between the sexes, this being a frequent adverse reaction (38%)16, while another, carried out on a sample of elderly people receiving treatment with doses of 12.5 to 300 mg/day of trazodone found no association with QT17 interval prolongation. Importantly, trazodone has been shown to increase the risk of suicidal ideation, not only in patients depressed. A study conducted to compare the safety profile of medications for the treatment of insomnia in adults showed that subjects taking trazodone (< 200 mg) were at risk of ideation greater suicidal compared to those who took zolpidem (HR 1.61 CI 1.07-2.43). There were no statistical differences in terms of suicidal ideation between zolpidem and other drugs with a sedative effect (benzodiazepines, antihistamines).
Other adverse effects that have been reported very rarely are: priapism (0.1-0.01%), increased libido, gastric upset, nausea, vomiting, loss of appetite, bleeding, hyponatremia, acute angle glaucoma, and mania/hypomania (3). As with other antidepressants, at the end of treatment a reduction is recommended gradual dosing to avoid or minimize the appearance of a withdrawal syndrome.
PLACE IN THERAPEUTIC
Insomnia is one of the psychiatric disorders that Primary Care professionals encounter more frequently in their daily clinical practice. The predominant symptom is difficulty initiating or maintaining sleep, or not having a restful sleep, for at least a month. This disturbance causes clinically significant discomfort, as well as social, work or other areas of the individual’s life deterioration. Your treatment is based on three pillars fundamentals: sleep hygiene, psychological interventions and pharmacological treatment. Although the therapies cognitive-behavioral are the treatment of choice, the prescription and the use of drugs for insomnia is very widespread.
Benzodiazepines and non-benzodiazepine hypnotics (zolpidem, zopiclone, zaleplon) are the drugs of first line, although they are recommended in the short term and at the lowest possible dose, due to their reaction profile and the risk of developing tolerance and dependence, as well as withdrawal syndrome. Therefore, it has spread the use of other medications, such as tricyclic antidepressants, melatonin, or trazodone. Trazodone is an antidepressant with a sedative effect, which is used in our country not only for depression, but also for also in indications not yet covered in its data sheet, such as insomnia. Doesn’t seem to change importantly the architecture of sleep and has low potential to develop dependency, aspects that attractive for use, especially in the elderly, in whom benzodiazepines and Z-hypnotics have multiple drawbacks.
However, the available evidence of its efficacy in this indication is scant, and comes mainly from trials Small, short-term clinical trials in patients with depressive disorders. On the other hand, precisely because of the scarcity of studies carried out for the treatment of insomnia not associated with depression, since the doses used in them are lower than those recommended as an antidepressant, no conclusions can be drawn about its benefit-risk balance.
Most of the guidelines consider that the evidence is insufficient to recommend it in the general population or in the elderly with chronic insomnia. The use of antipsychotics or sedating antidepressants (such as trazodone) only it is justified when there is a concomitant pathology. It could be a therapeutic option in patients who do not.
They respond well to first-line drugs. Its use in monotherapy is recommended, at lower doses than those commonly used for the treatment of depression and for short periods or intermittently, taking into account the characteristics of the patient1. Finally, it is opportune to remember that the use of trazodone or any medication in different conditions to those authorized in the data sheet (different indications, dosage, population group, etc.) requires compliance with the conditions established by Royal Decree 1015/2009, which regulates the availability of medicines in situations specials. For use outside the technical data sheet, which must be exceptional, the doctor must properly justify in the clinical history the need for the use of the drug, report suspected adverse reactions, inform to the patient in understandable terms of the nature of the treatment, its importance, implications and risks, and obtain their informed written consent. The fact that off-label use of a medication does not exempt from compliance with this regulation.
CONCLUSIONS
- The scientific evidence available on the use of trazodone for insomnia is limited, being mostly part of clinical trials with few patients, generally with underlying depressive disorders.
- The main clinical practice guidelines do not recommend it as a treatment for insomnia, especially long-term term.
- Its use can be considered when cognitive-behavioral therapies have not been effective or the drugs are poorly tolerated. hypnotics, taking into account the characteristics of the patient, for a short period of time and at low doses.
- Due to its safety profile, special caution should be taken in the elderly and patients with cardiac pathology.
- The use of trazodone for insomnia is not among the indications on the data sheet, so requires compliance with regulatory requirements (exceptional use, justification in the medical record and request for informed consent to the patient)
How to purchase trazodone – article.
Written by Andrew Scott, MD. 08/22/2023